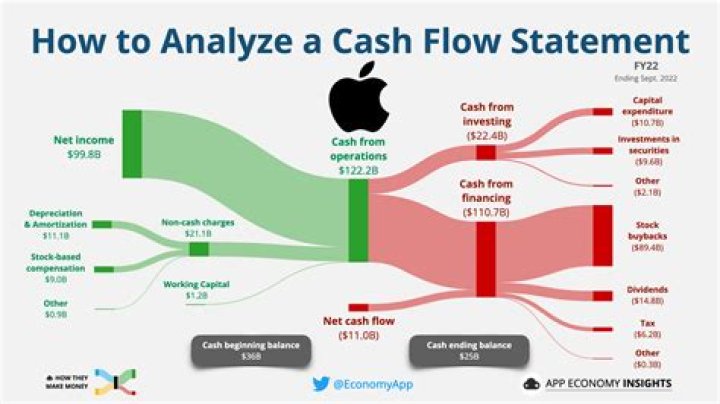
Which element has higher electron affinity F or Cl?

Joseph Russell
Updated on May 07, 2026
Also asked, does F or Cl have higher electron affinity?
Electron affinity of fluorine is less than that of chlorine. This is due to the reason explained below: Fluorine has seven electrons in 2p-subshell whereas chlorine has seven electrons in its 3p-subshell. Therefore, repulsion among the electrons will be more in the 2p-shell of fluorine than 3p-subshell in chlorine.
Additionally, which F Cl has the highest electron affinity and why? Now coming to the question, as we have already seen; Br has the lowest electron affinity because of high atomic size and therefore has less tendency to accept electrons to form anion whereas Cl has highest electron affinity because of less atomic size and has high tendency to accept electrons.
In this regard, which is more electronegative F or Cl?
At the bottom left corner of the periodic table lies Cesium, which lacks a catchy nickname [but has attracted a strangely devoted following] with an electronegativity of 0.79.
Carbon is More Electronegative Than You Think.
| Element | Electronegativity (Pauling) |
|---|---|
| F | 4.0 [3.98] |
| O | 3.4 [3.44] |
| Cl | 3.2 [3.16] |
| N | 3.0 [3.04] |
Does CL have electron affinity?
The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous -1 ions. First electron affinities have negative values. For example, the first electron affinity of chlorine is -349 kJ mol-1. By convention, the negative sign shows a release of energy.